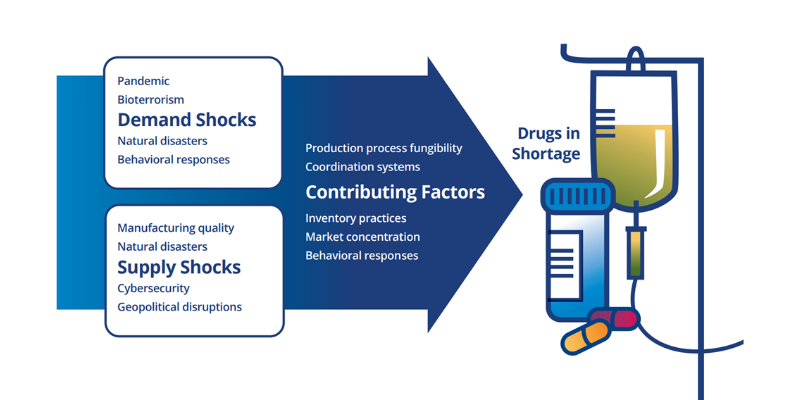
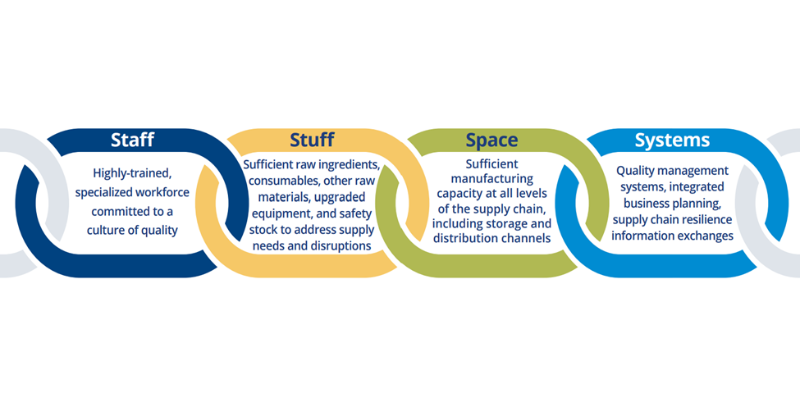
Duke-Margolis ReVAMP Drug Supply Chain Consortium

About the Consortium
ReVAMP Drug Supply Chain Consortium Members
Advisory Group
|
Laura Bray |
Nicolette Louissaint Senior Vice President, Policy and Strategic Planning Healthcare Distribution Alliance |
|
Angie Boliver |
Gerren McHam Director of Government and External Affairs API Innovation Cente |
|
Hilary Daniel |
Leigh Purvis |
|
Dave Ehlert |
Whitley Quan Director, Head of U.S. Public Policy Cencora |
|
Celeste Frankenfeld Lamm |
Stephen Schondelmeyer Professor and Director, PRIME Institute, Department of Pharmaceutical Care and Health Systems University of Minnesota |
|
Michael Ganio |
Eric Tichy Division Chair, Pharmacy Supply Solutions; Associate Professor of Pharmacy Mayo Clinic |
|
John Gray |
Michael Turchetti |
|
Vadim Gurvich |
Terri Lyle Wilson |
Observers
|
Trinidad Beleche |
Tracey Koehlmoos |
|
Brooke Courtney |
Jennifer Maguire Director, Officer of Pharmaceutical Quality/Office of Quality Surveillance U.S. Food and Drug Administration |
|
Adam Fisher |
Alana Moore Policy Analyst U.S. Drug Enforcement Administration |
|
Arlene Joyner |
Ted Oja |
|
Gregory Kavanagh |
Manuel Osorio Lead, CBER Advanced Technologies Team U.S. Food and Drug Administration |
|
|
Interested in joining the effort? For membership inquiries, please contact Stephen Colvill and Thomas Roades.
Duke-Margolis Project Team Leads

Stephen Colvill, MBA
Assistant Policy Research Director

Marianne Hamilton Lopez, PhD, MPA
Senior Research Director, Biomedical Innovation
Faculty Director of the Duke-Margolis Postdoctoral Associates & Affiliated Fellows Program
Adjunct Associate Professor
Senior Team Member
Margolis Core Faculty

Thomas Roades, MPP
Policy Research Associate

Madi Cordle
Policy Analyst, Assistant
Chan Harjivan
Visiting Fellow